Difference between Trans and Cis Resveratrol
By now, you’ve probably heard about resveratrol and its magical health-bestowing properties. But do you know that there are two forms of 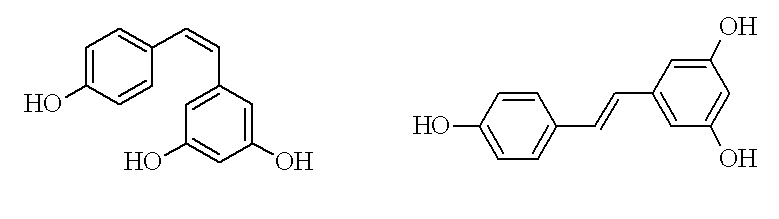 resveratrol? Trans and Cis resveratrol. You’d also have noticed that resveratrol supplements have trans-resveratrol printed on their packaging.
resveratrol? Trans and Cis resveratrol. You’d also have noticed that resveratrol supplements have trans-resveratrol printed on their packaging.
Do you know what these terms mean? Do you know which form of resveratrol is good for you?
Well, here are some answers. But you must be okay with basic chemistry to understand these terms. Anyway, here goes:
Every compound or molecule on Planet Earth, just like our resveratrol, contains at least two atoms that are chemically bonded to each other. For example, water is represented by the chemical formula H2O, which means 2 parts of hydrogen and 1 part of oxygen have bonded together to make one molecule of water; Likewise, the sugar molecule is represented by the formula C6H12O6 – which means that one sugar molecule is made up of six carbon atoms, 12 hydrogen atoms and 6 oxygen atoms.
Just like in the examples above, every molecule is represented by a chemical formula. Some formulas are simple, some are complex. What you have to understand is that science has created a method to name each formula.
Now, these chemical formulas are as per the IUPAC (International Union of Pure and Applied Chemistry) system.
Resveratrol’s chemical formula is C14H12O3 (14 atoms of carbon, 12 atoms of hydrogen and 3 atoms of oxygen) and its chemical name is 3,5,4'-trihydroxy-trans-stilbene.
The IUPAC system states when two important molecules contained in the main molecule face each other, then the main molecule is “cis.” When two important molecules face away, then the main molecule is “trans.”
Chemical Formula of Cis & Trans Resveratrol...Now, Check resveratrol’s diagram above. You will observe that 2 important molecules are together in cis resveratrol (on your left) and apart in trans resveratrol (on your right). To learn a little more as to who discovered resveratrol you need to read through the following page above as it will give you a good overview on just how long this product has been available.
This is the description of cis and trans resveratrol.
Now you may think that resveratrol is resveratrol – what difference does it make how the molecules are located inside resveratrol?
Well, this is chemistry not mathematics. In mathematics, 3 + 2 and 2 + 3 equal and mean the same thing. But when we are talking molecules and compounds, then the positioning of each major molecule within the main molecule can make a difference. Different positioning of molecules can have different effects.
Now, let’s understand the difference between trans and cis resveratrol.
Laboratory experiments conducted by both medical and scientific researchers categorically state that trans resveratrol is effective and powerful, while cis resveratrol is not effective. They opine that trans resveratrol is stable and can be easily absorbed by the human body, while cis resveratrol is not as stable and the human body cannot absorb cis resveratrol easily.
Going to the how and why of why trans resveratrol is better than cis resveratrol is way beyond the scope of this article. But you don’t have to bother about that because it involves some very intense and complex science, which can get very boring and leave your brain in a fog.
So, if you have decided to take resveratrol supplements, go with trans resveratrol because of the reasons specified above.
You now know the description and difference between trans and cis resveratrol, you know how each resveratrol is chemically bonded, and you know which resveratrol works better. So, go on ahead and approach resveratrol with confidence, for now, you are an informed buyer.
